Live
- Turkish police detain 11 suspected IS members
- Vice FMs of South Korea, Canada discuss cooperation in energy, G7 ties
- HUL posts 6 pc fall in Q4 net profit at Rs 2,406 crore, declares dividend of Rs 24 per share
- Zimbabwe name 15-member squad for five-match T20I series against Bangladesh
- ‘I would love to see more players from my state get opportunities,’ I-League Emerging Player Gyamar Nikum
- More women voters in 14 Karnataka LS seats which go to polls on May 7: CEO
- YSRCP Candidate for Vijayawada West, Assures Voters of Inclusive Leadership
- Buy iPhone 15 at Rs 62,999 on Flipkart with Bank Discounts
- Kandikunta Venkataprasad Intensifies Election Campaign in Tribals Areas of Sri Sathya Sai District
- Kapusankshema Seva Sangam Extends Support to TDP-BJP Alliance as per Janasena Chief's Instructions
Just In
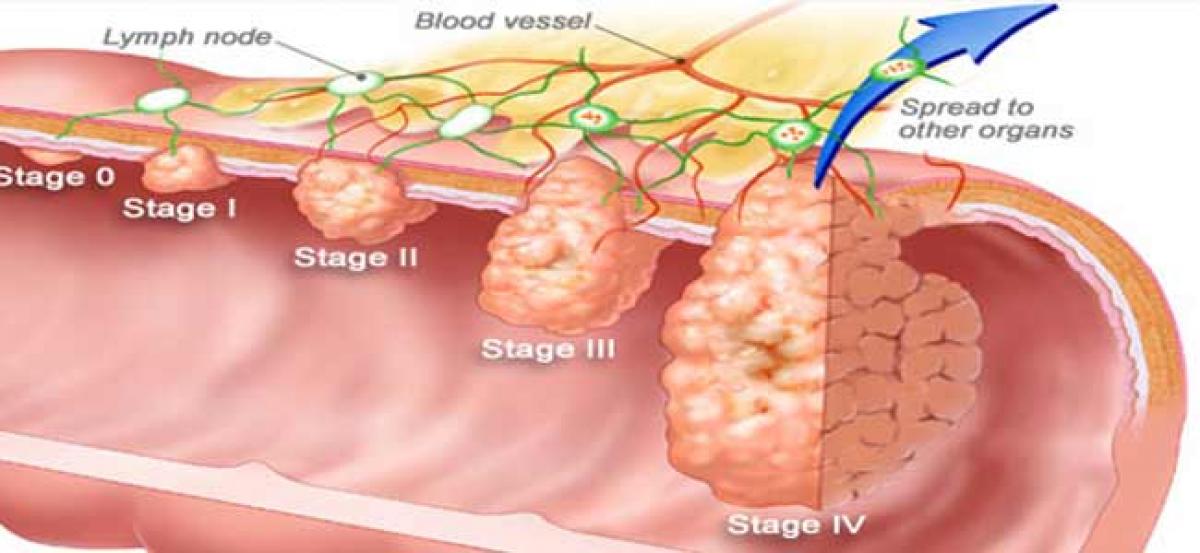
Colorectal cancer encompasses both cancers of the colon and the rectum. According to the World Health Organisation (WHO) and the Centers for Disease Control and Prevention (CDC), it is the second-most common cancer worldwide after lung cancer.
Cancer is a terrifying disorder which has touched almost everyone in some way or the other. With incidents of cancer rising across the globe, scientists have been frantically researching to figure out solutions to treat this disease in the most effective and non-invasive manner.
Colorectal cancer encompasses both cancers of the colon and the rectum. According to the World Health Organisation (WHO) and the Centers for Disease Control and Prevention (CDC), it is the second-most common cancer worldwide after lung cancer. In general, most colorectal cancers begin as a form of polyp. Some of these polyps can become cancerous over a period of years. The usual symptoms of colorectal cancer are diarrhea, constipation, blood in the stool, abdominal pain and bloating, fatigue and unexplained iron deficiency in men.
There are several factors which can increase the risk of an individual for colorectal cancer. Several lifestyle-related factors have been linked to an increased chance of getting colorectal cancer such as obesity, lack of physical activity, certain types of diets, especially those including highly-processed meat, smoking and alcohol intake.
In addition, certain medical conditions such as personal history of inflammatory bowel diseases, family history of colorectal cancer or adenomatous polyps and an inherited syndrome such as Lynch Syndrome can predispose an individual to developing colorectal cancer.
Science and medicine have made rapid advancements in colorectal cancer detection and treatment. Today, colorectal cancers are treatable and most of the patients have been known to lead a very normal life post treatment. At the same time, disease monitoring for such patients is a huge challenge. Carcinoembryonic antigen (CEA) is a routinely used biomarker for the surveillance in colorectal cancer patients. However, it is not a reliable marker.
In recent years, the blood-based liquid biopsy approach for monitoring colorectal cancer is being recommended. The pathology lab can extract tumor DNA from the blood sample monitoring disease progression based on the genetic mutations that are detected. Liquid biopsies are used to detect circulating tumour cells or circulating tumour DNA (ctDNA) to screen (non-invasively) for early-stage cancers, detect residual disease, or monitor response to treatment. Oftentimes, liquid biopsies can help explain why some cancers are resistant to certain therapies and better inform changes in treatment in response to the tumor's changing mutation profile.
With rising cancer incidence in India, including many colorectal cancers, it is crucial to continue to develop newer ways and methods to detect colorectal cancer early, increasing the success rate of treatment. In various studies conducted in a group of Indian colorectal patients, it has been found that there is significant evidence of the mutations of the KRAS (Kirsten rat sarcoma) virus and APC (adenomatous polyposis coli), a multi-functional tumor suppressor gene, co-existing in these patients. These mutations can be detected using a liquid biopsy approach providing clues towards disease progression.
To address the surveillance of colorectal cancer in treated patients and to study the efficacy of the treatment regimen a liquid biopsy approach is under development and will be available soon to doctors and patients in India. It offers key advantages over traditional solid tissue biopsies as it is a minimally invasive blood test. It also makes the analysis and the monitoring of the disease a lot easier as the need for the tissue biopsy gets eliminated completely.
It is essential to remember that colorectal cancer is treatable and genetic analyses can guide the treatment involving targeted therapy based routes and impact of surgery along with monitoring the disease.

© 2024 Hyderabad Media House Limited/The Hans India. All rights reserved. Powered by hocalwire.com







