India's vaccine wait is over
India’s vaccine wait is over
DCGI clears Oxford’s Covishield, Bharat Biotech’s Covaxin
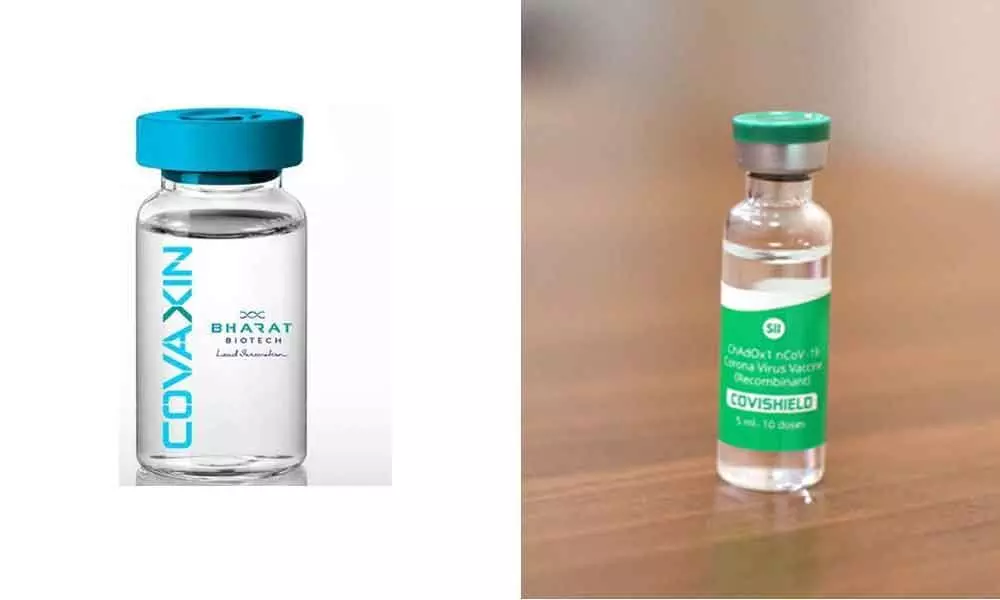
New Delhi: Two vaccines for coronavirus, Oxford Institute's Covishield, developed by the Serum Institute and Bharat Biotech's Covaxin, received emergency approval from the country's drug regulator on Sunday.
"We'll never approve anything if there is slightest of safety concern. The vaccines are 110 per cent safe," Drug Controller General of India VG Somani said, adding that Covishield was found to be 70.42 per cent effective and Bharat Biotech's Covaxin was "safe and provides a robust immune response".
The Serum Institute of India (SII) has already stockpiled some 80 million doses of Covishield and, as such, the rollout can begin fairly quickly. However, the rollout of Covaxin, being manufactured by Hyderabad-based Bharat Biotech in collaboration with the Indian Council of Medical Research (ICMR), could take a few days or weeks to be available.
What next?
First, the entire vaccination drive will be voluntary. The government has already announced that first in line will be 3 crore workers in the forefront of India's battle against the novel coronavirus, including 1 crore healthcare workers and 2 crore frontline workers.
Union Health Minister Dr Harsh Vardhan said on Saturday that the vaccine will be administered to them for free. Also receiving the vaccine in the first phase will be a third priority group – consisting of some 27 crore persons above age 50, and persons below age 50, but with associated comorbidities.
The government aims to complete the first phase of vaccinations by August 2021. The timelines for the rest of the population are not known as yet.
However, it is not as though the rest of the population will have to wait until the first phase of vaccination is complete. Other groups will begin to be inoculated simultaneously after some weeks or months.
Mock drills
Preparations have been on for weeks for what will be India's widest and most ambitious vaccination drive ever. Two rounds of mock drills have already been held — the first in four states on December 28-29, and the second on January 2, covering 285 session sites in 125 districts in all states around the country.
Some 96,000 vaccinators have been trained, including 2,360 in a National Training of Trainers, and over 57,000 in district-level training carried out in 719 districts.
More than 75 lakh beneficiaries have been registered on the Co-WIN platform, which will provide real-time information on vaccine stocks, their storage temperature, and individual beneficiaries of the shots, Dr Harsh Vardhan said on Saturday.